COPYRIGHT © 2016-2026. The Owl Teacher | Privacy page | Disclosure Page | Shipping | Returns/Refunds
Engage with Holiday Science
Every year without fail, as we get closer and closer to that big starting date of winter break, students become more and more restless. I have yet to have a class that didn’t. And to be honest, I have yet to feel differently myself. After Thanksgiving, I, too, am counting down the days before I can wear pajamas around the house and just kick back and relax a few days. Students and teachers alike need that winter break as a time to regroup. But getting to that time is the tricky part… Or is it? We can get there a little faster with some holiday science!

My students love science. I think it is because they can really get hands-on with it and because, to some degree, it’s a form of “magic” to them. So, I decided to “hold their attention” this year before winter break. I was going to play along with the Christmas or Holiday spirit and mesh it with a little science – and to be punny – yes, punny (like pun) – I’d call it, “The Twelve Days of Christmas Science!” because really, it was a holiday science, right?
I put together 12 different activities that are holiday- and science- (get it, holiday science?) related that I think my kiddos will enjoy. First, I needed to test them all out at home–plus, it creates a little fun bonding (play!) time with my own children. (I always test out anything science-related at home before in the classroom or before creating science resources so I KNOW it works!)
With that said, forgive the home environment in the background. On the first day of Christmas, my true love gave to me…the strength of a candy cane. Just how much weight can a candy cane hold? And is the hook stronger or the straight side? I just had to know – and I had my own theories.
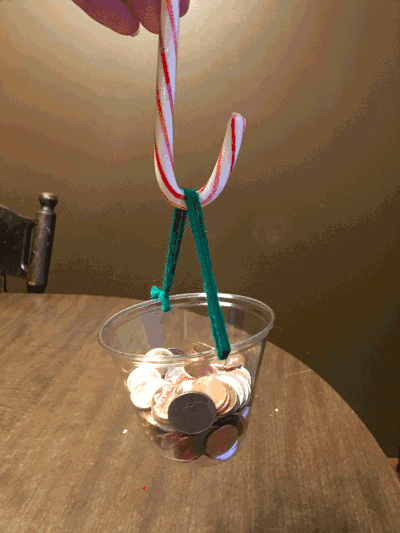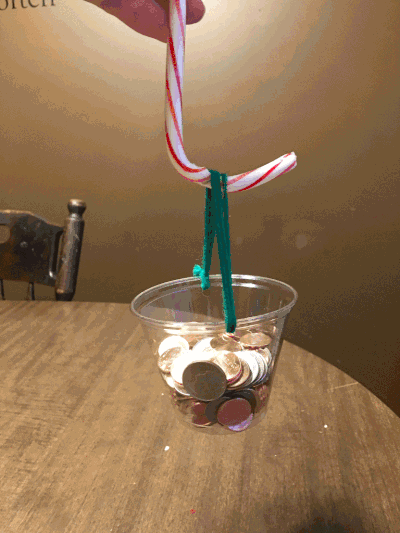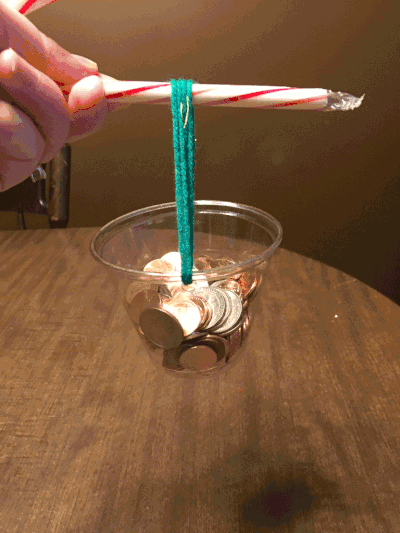
On the second day of Christmas, my true love gave to me…Christmas lights! Students love to play with electricity. So, I thought they could explore how many lights they can light up on one single 9V battery, and we could use this as an opportunity to discuss open and closed circuits, along with parallel and series circuits.
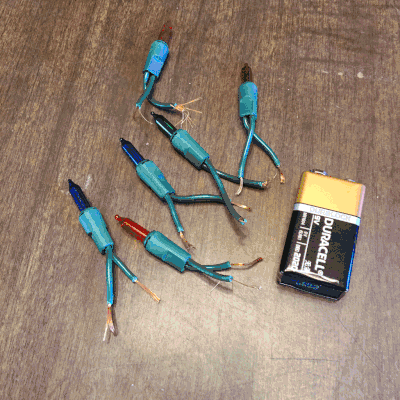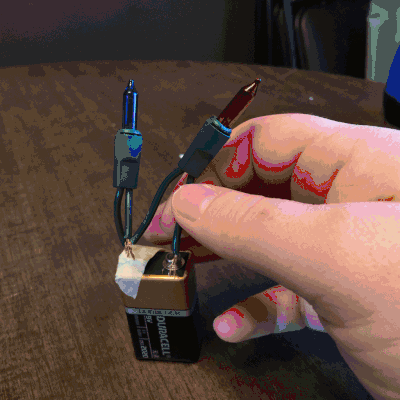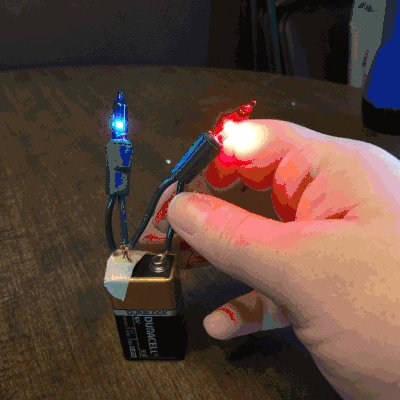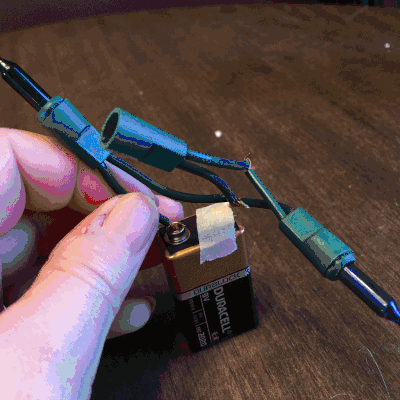
On the third day of Christmas, my true love gave to me…dancing reindeer droppings! Oh, yessss! I just know the name alone will get my kiddos cooooing! I will admit, when I first set up this holiday science demonstration, I was expecting the raisins to go wild. They really don’t, but it was fun, nonetheless.
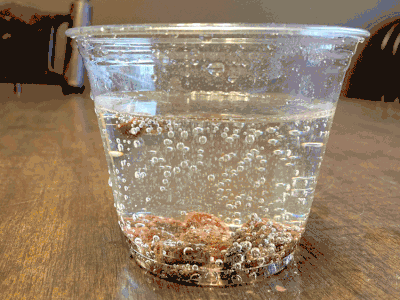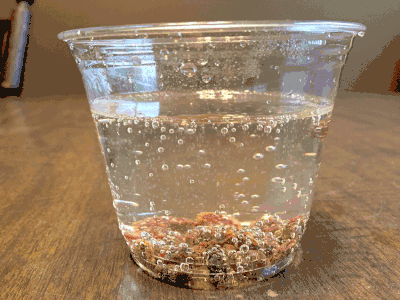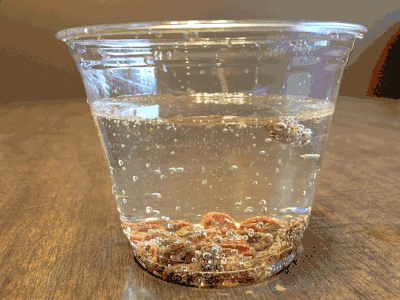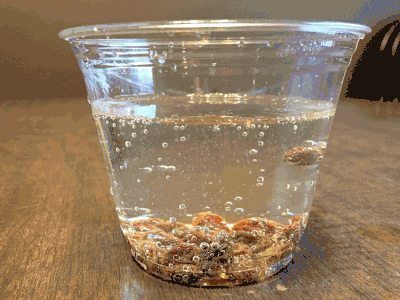
On the fourth day of Christmas, my true love gave to me…Santa boogers (red slime). Sorry. No pictures. I have made that so many times I could probably make it in my sleep. And you just can’t make slime around three-year-old twins, or you can say bye-bye to your nice carpet. I know the kiddos will LOVE it, and so will the parents. 😉
On the fifth day of Christmas, my true love gave to me…candy cane testing. In this engaging activity, students will determine which dissolves faster: a round peppermint candy or a candy cane. And, does it matter if the candy cane is broken up? (And do they weigh the same?) Lots to ponder!

On the sixth day of Christmas, my true love gave to me…reindeer treats! Yum! Mixtures and solutions can definitely be confusing for students, so why not have them practice and enjoy a treat? I threw a little reindeer food in a bag and created a yummy drink. I think the students will have to decide which is a mixture and which is a solution. Then, they’ll have to tell me how they know!

On the seventh day of Christmas, my true love gave to me…present time! Who doesn’t love presents? I know students will enjoy this activity because it involves chocolate. I had to test for a while (and of course eat the mistakes!) to find the right combination of treats that float and those that don’t. When we’re done, I think we can talk about buoyancy and density! (And eat our presents!)

On the eighth day of Christmas, my true love gave to me…droppings! Well, we had previously discussed reindeer droppings and density, so let’s combine them for a little fun. You see Rudolph and Frosty the Snowman were both having a conversation about…well, you know. So, we decided to test things out.

We can go back to discussing all those liquids on one another and why that raisin sank to the bottom and then just hung out on top of the bottom layer (corn syrup). I love density layers. Another thing: Don’t add as much food coloring as I did. I went overboard.
On the ninth day of Christmas, my true love gave to me…a disappearing Frosty! I can’t resist. I just love watching things be destroyed…well, the rights things, I should say. Haha. It’s my son’s fault. He’s rubbed off on me.
On the tenth day of Christmas, my true love gave to me…candy chromatography! This one was tricky for sure. I learned two very important things.
- I hate coffee filters for this process, so I switched to paper towels.
- You really, really must let the candy cane dissolve to a high concentration in the drop of water before you start. However, I still was able to see it work and know for when I’m in the classroom! No awesome animation…

On the eleventh day of Christmas…and on the twelfth day of Christmas…
Well, they are top secret! (Don’t you just love it when people do that?)
But, you can find out the top secret information by heading to my Teachers Pay Teachers store and picking up my Twelve Days of Christmas Science packet to complete in your classroom! All of the above experiments (with better detail and engaging wording for your students) are in it! It’s a great way to keep your students engaged and to explore holiday science – AND it saves you time and energy from having to whip this all together yourself.

Now, even if you don’t feel confident teaching science, these activities are easy for any teacher because they include background knowledge and everything you need to be successful (except the actual physical materials).
And because you are my true loves (fans/followers/supporters), I have for you a special freebie! You can try this experiment out and see what you think!
You’ve seen this one before, right?
What would the reaction be of a candy cane placed in baking soda?

Where did the candy cane go?
What would the reaction be of a candy cane placed in water?
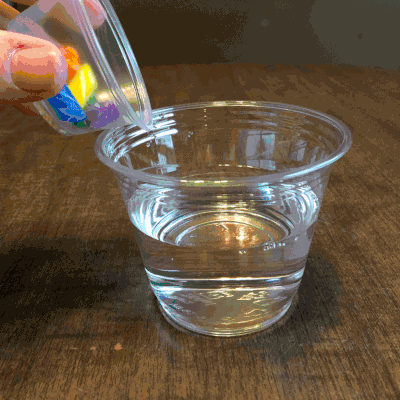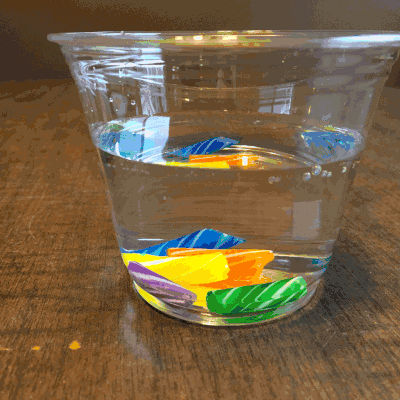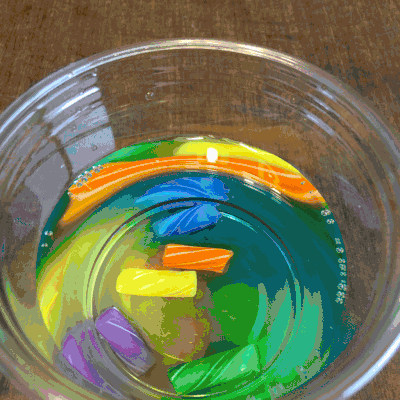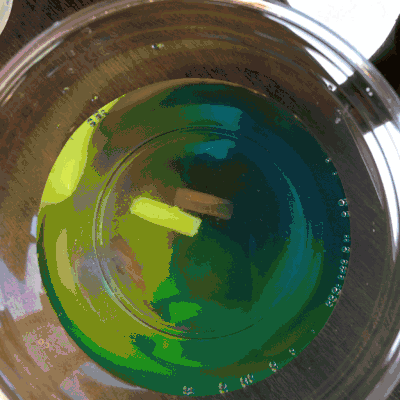
Colorful, but boring, am I right?
What about a candy cane placed in club soda?
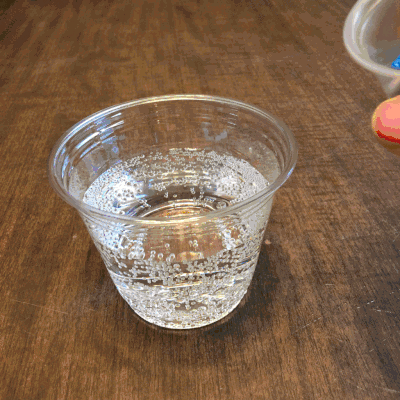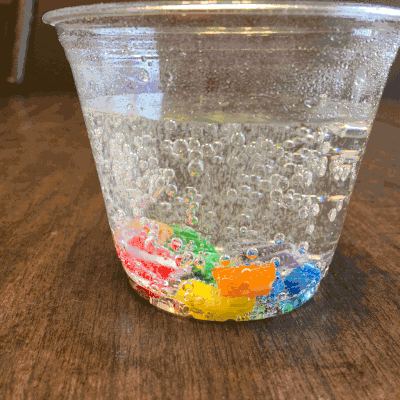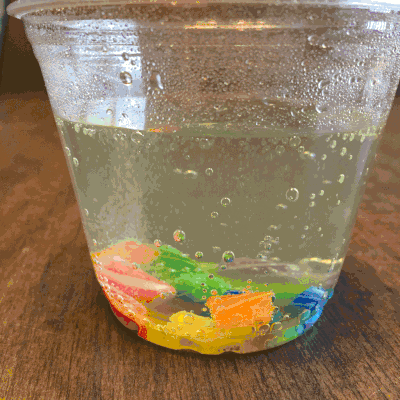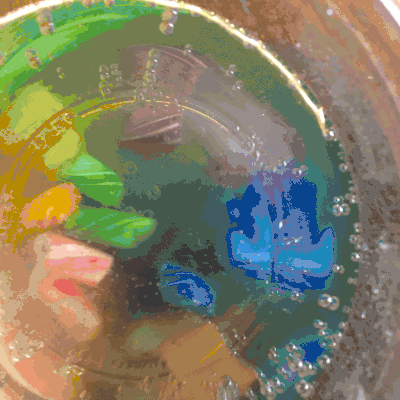
Now it’s starting to get interesting…bubbling.
But… What about a candy cane placed in B-L-E-A-C-H?
I am crazy. I was doing this thinking that this would easily strip away the dyes in the candy cane. At first, I got nothing. So I set it aside and started playing around with other things while I waited *yawn* for it to start dissolving… Then, all of a sudden, out of NOWHERE… WHAM! It went berserk! I mean, bubbles everywhere – and high! Loud popping, horrible smell, the cup started melting, and when it was done, the candy cane looked like it was chewy. I had to ventilate my house quickly! I don’t recommend doing that one in your classroom, at least not without doing it in your own house first and seeing what you think. I wouldn’t do it. I still don’t know what happened, besides a chemical reaction. They were just regular ol’ candy canes. I questioned if I was going to live to write this post. 🙂

GRAB YOUR FREEBIE!
Anyway, the freebie below is just an open-ended sheet where you can experiment with placing candy canes in different substances to see what the reaction will be. I’d recommend playing around with a variety of candy canes, too – sour ones, tart ones, peppermint ones, break them up, keep them whole – it doesn’t matter, as long as you’re having fun! You can get this Holiday Science Freebie (below) by clicking here and downloading it free!

And (yes, I have started a lot of sentences with “and” today…I’m still in Thanksgiving break mode), before I go, my husband insisted that I include this video — because when I start playing with science experiments around the house, he has to get involved. (If you don’t believe me, see my Teaching Electricity with Pickles Post.)
So, consider this a challenge. Have your students see if they can replicate it.
Make the “North Pole” – that’s what we will call it! Ha!
If you enjoyed any of these science activities, then please consider following my Teachers Pay Teachers store, where you will find a vast amount (well, not a vast amount, but a lot) of science units and activities that are hands-on and engaging!
Other related products you may enjoy!
Head to my TpT Store to purchase the 12 Days of Holiday Science today!